A13 - Golgi-associated protein scaffolds regulating receptor targeting and hormone secretion
Principal Investigator
Prof. Dr. Annette Schürmann, DIfE
Hormone secretion and the targeting and dynamics of their respective receptors are crucial for the function of metabolically active tissues and organs such as adipose tissue and the pancreas. Work by us and others has identified critical roles in these processes for Golgins, large coiled-coil scaffolding proteins that act by orchestrating vesicle-mediated protein traffic in and out of the Golgi complex. In the past funding period we have demonstrated that the GTPase ADP-ribosylation factor related protein 1 (ARFRP1) recruits the GRIP domain-containing Golgins Golgin-97 and Golgin-245 to the trans-Golgi network (TGN), a process required for regulating lipid droplet protein composition in intestinal cells (Werno et al., 2018) , recycling of the transferrin and insulin receptors (Rödiger et al., 2018) and for hormone secretion (Stadion et al., 2018).(Figure 1)
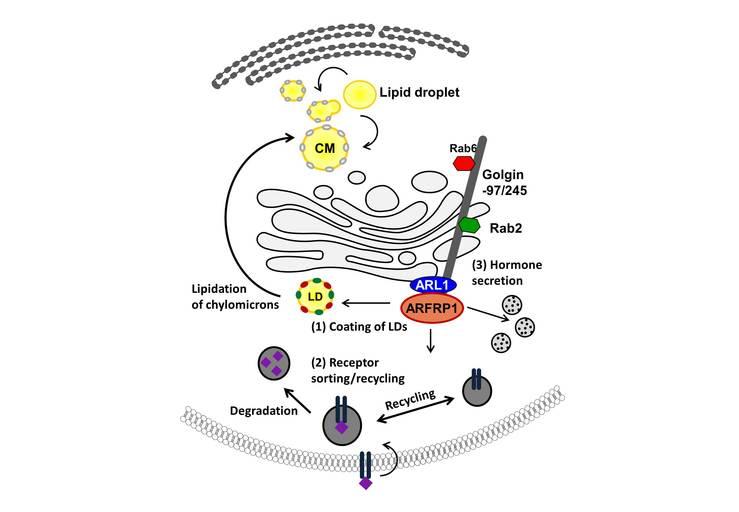
Figure 1: Schematic representation of ARFRP1 mediated scaffolds and their respective functions. ARFRP1 function is required for (1) the protein composition of LD coats to regulate lipolysis and lipidation of chylomicrons in the intestine, (2) correct sorting and recycling of the insulin receptor in adipose tissue, and (3) hormone secretion from adipocytes (adiponectin) and pancreatic beta-cells (insulin).
Within project A13 we further investigate the molecular mechanism of ARFRP1 dependent trans-Golgi scaffolds in metabolically active tissues and organs like skeletal muscle and the pancreas. We use different cell biological and biochemical methods as well as tissue specific KO mouse models. Furthermore, we take advantage of the bioimaging facility within our consortium to unravel the subcellular distribution and dynamics of the proteins we study. The goal of the current funding period is to functionally characterize new identified ARFRP1 interacting proteins and the biological means of this molecular interplay.
References:
-
Kluth, O., M. Stadion, P. Gottmann, H. Aga , M. Jähnert, S. Scherneck, H. Vogel, U. Krus, A. Seelig, C. Ling, J. Gerdes & A. Schürmann. Decreased Expression of Cilia Genes in Pancreatic Islets as a Risk Factor for Type 2 Diabetes in Mice and Humans. Cell Rep. 26,3027-3036 (2019)
- Sztul ,E., P.W. Chen, J.E. Casanova, J. Cherfils, J.B. Dacks, D.G. Lambright, F.S. Lee, P.A. Randazzo, L.C. Santy, A. Schürmann, I. Wilhelmi, M.E. Yohe & R.A. Kahn. ARF GTPases and their GEFs and GAPs: concepts and challenges. Mol Biol Cell. 30,1249-1271 (2019)
- Rödiger, M., M.W. Werno, I. Wilhelmi, C. Baumeier, D. Hesse, N. Wettschureck, S. Offermanns, K. Song, M. Krauß & A. Schürmann. Adiponectin release and insulin receptor targeting share trans-Golgi-dependent endosomal trafficking routes. Mol Metab. 8,167-179 (2018)
-
Stadion, M., K. Schwerbel, A. Graja, C. Baumeier, M. Rödiger, W. Jonas, C. Wolfrum, H. Staiger, A. Fritsche, H.U. Häring, N. Klöting, M. Blüher, P. Fischer-Posovszky, T.J. Schulz, H.G. Joost, H. Vogel & A. Schürmann. Increased Ifi202b/IFI16 expression stimulates adipogenesis in mice and humans. Diabetologia. 61, 1167-1179 (2018)
-
Werno, M.W., Wilhelmi, I., B. Kuropka, F. Ebert, C. Freund & A. Schürmann. The GTPase ARFRP1 affects lipid droplet protein composition and triglyceride release from intracellular storage of intestinal Caco-2 cells. Biochem Biophys Res Commun. 506, 259-265 (2018)