A07 - Regulation of SH3 domain-based scaffolds in clathrin-mediated endocytosis
Principal Investigators
Prof. Dr. Christian Freund, FU Berlin
Prof. Dr. Volker Haucke, FMP Berlin
In project A07 the SH3 domain containing scaffolding proteins intersectin 1, endophilin A1 and SNX9 and their interaction partners are studied in molecular detail. These proteins are critical for endocytosis and/or synaptic vesicle clustering and the goal here is to understand the mechanistic prerequisites of the compositional dynamics that accompanies synaptic vesicle cycling. Since proline-rich sequence (PRS) recognition by SH3 domains is characterized by interactions with high off-rates, dynamic changes in macromolecular complex associations can be rapidly tuned by the abundance, the localization and the post-translational modifications of the SH3 domains or their interaction partners. We could show that intersectin 1 and endophilin directly interact by an unusual SH3-SH3 domain interaction, still allowing proline-rich sequences, for example from dynamin, to bind to the canonical endophilin-SH3 binding site (Fig. 1). 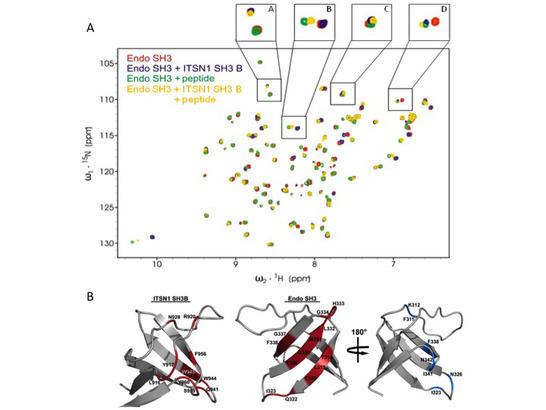
Figure 1: Intersectin 1 (ITSN-1) SH3 B domain and endophilin A1 SH3 domain directly interact. A, Superposition of NMR spectra of the isolated Endophilin A1 SH3 domain (red), the domain in presence of ITSN1 SH3 B domain (blue), in presence of peptide (green) or in the presence of both interaction partners (yellow). Analysis of the data shows that both interaction partners can bind at the same time. B, Epitope mapping model of intersectin 1 SH3B domain and of Endophilin A1 SH3 domain, when bound to each other. Note that the endophilin A1 SH3 epitope comprises a non-canonical b-sheet and not the typical nSrc and RT loop epitopes. The proline-rich ligand binding site (blue colors in the lower right panel) is independent of this b-sheet binding site for intersectin 1.
In the current funding period we plan to further investigate SH3 domain mediated interactions in the larger complex and in the context of lipid membranes. Furthermore we will investigate the interplay between intra- and intermolecular interactions by structural biology and biophysical methods.
References:
- Pechstein, A.., Gerth, F., Milosevic, I., Jäpel, M., Eichhorn-Grünig, M., Vorontsova, O., Bacetic, J., Maritzen, T., Shupliakov, O., Freund, C. & Haucke, V. Vesicle uncoating regulated by SH3-SH3 domain-mediated complex formation between endophilin and intersectin at synapses. EMBO Rep.16, 232-239 (2015)
- Posor Y, Eichhorn-Gruenig M, Puchkov D, Schöneberg J, Ullrich A, Lampe A, Müller R, Zarbakhsh S, Gulluni F, Hirsch E, Krauss M, Schultz C, Schmoranzer J, Noé F, Haucke V. Spatiotemporal control of endocytosis by phosphatidylinositol-3, 4-bisphosphate. Nature 499, 233-237 (2013)
- Saheki, Y. & De Camilli, P. Synaptic vesicle endocytosis. Cold Spring Harbor Perspec Biol 4, a005645-a005645 (2012)
- Pechstein, A., Shupliakov, O. & Haucke, V. Intersectin 1: a versatile actor in the synaptic vesicle cycle. Biochem. Soc. Trans. 38, 181-186 (2010)